Get a quoate
What is Vaporized Hydrogen Peroxide (VHP) Sterilization? A Complete Guide
Published : 2026-04-15Unlike steam sterilization where the D-value remains relatively stable, the D-value during Vaporized Hydrogen Peroxide (VHP) sterilization fluctuates significantly based on concentration, humidity, and condensation levels. Consequently, a unified calculation like F0 is not applicable. Based on USP <1229.11> “Vapor-Phase Sterilization” and relevant research, this article explores the definition and the four critical phases of the VHP process.
Q: What is Vaporized Hydrogen Peroxide (VHP) Sterilization?
A: According to USP <1229.11>, VHP sterilization is a process where liquid hydrogen peroxide (H₂O₂) is flash-vaporized into a gas phase and delivered into an enclosed space (such as isolators, cleanrooms, or equipment). By forming controlled micro-condensation on surfaces, it achieves microbial inactivation (including spores) through strong oxidation, resulting in effective sterilization or decontamination.
- Vapor Phase: H₂O₂ is liquid at room temperature; it is only referred to as “vapor phase” after vaporization.
- Sterilization: The ability to kill bacterial spores and achieve a Sterility Assurance Level (SAL) of ≤ 10⁻⁶.
- VHP: Abbreviation for Vaporized Hydrogen Peroxide.
Q: What are the Phases of the VHP Sterilization Process?
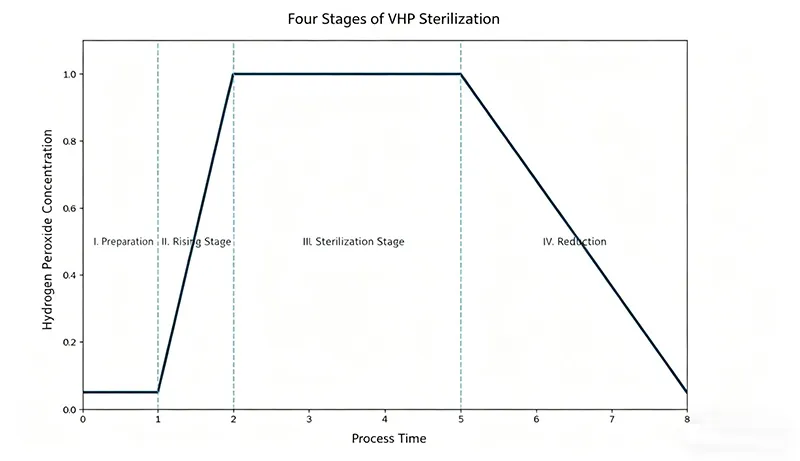
The cycle typically consists of the following four distinct stages:
I. Preconditioning Phase (Preparation)
This phase involves preheating and dehumidifying the equipment and piping to create a condensation-free environment for flash vaporization. Simultaneously, the target enclosure is dehumidified to prepare the surface for vapor contact.
II. Injection Phase
Liquid hydrogen peroxide is flash-vaporized into gas and delivered into the enclosed space via the HVAC air ducts and HEPA filters. During this stage, the H₂O₂ concentration rises steadily until it reaches the target setpoint.
III. Exposure Phase (Sterilization)
Once the concentration reaches the predefined limit, it is maintained at a steady state. This phase typically lasts 2 to 3 hours to ensure thorough microbial kill across all surfaces.
IV. Aeration Phase (Ventilation)
The system cools down while residual hydrogen peroxide is removed. Aeration continues until the concentration drops to a safe level of≤1 ppm (per U.S. OSHA Federal Regulation 29 CFR 1910.1000).
In the fields of modern biopharmaceuticals and high-tech manufacturing, selecting the right sterilization technology is just as important as establishing an appropriate production environment; indeed, one could argue that the two are indispensable. Many industry professionals are well aware that the advantages of the VHP (Vaporized Hydrogen Peroxide) process are clear—it reliably meets the surface sterility requirements of USP <1229.11>. However, to truly maximize these benefits, simply using the right process is not enough; the key lies in deeply integrating it with a well-designed, highly adaptable modular cleanroom. After all, even the most advanced sterilization technology will struggle to achieve the desired sterilization results without the support of a corresponding clean environment.
For facility managers, the core value of modular cleanrooms lies in their flexibility and practicality. With this layout, we can flexibly isolate critical production areas dedicated to VHP disinfection without disrupting normal operations in other parts of the facility—a solution that is both efficient and hassle-free. Whether you are responsible for sterility control in a small laboratory or cleanliness assurance for a large-scale filling production line, rapidly deployable modular cleanrooms, paired with a validated VHP disinfection cycle, work in tandem to help you find the optimal balance among production efficiency, regulatory compliance, and operational safety.
If you’re currently struggling to find a suitable solution for a customized modular cleanroom with integrated VHP decontamination capabilities, consider learning more about Suzhou Hansen. We specialize in customizing these solutions, providing end-to-end engineering services—from initial CAD design and on-site construction to final validation—to support you every step of the way with our professional expertise. Contact us today to receive personalized professional consultation and a free quote, helping you build a solid defense of cleanliness and sterility for your production environment.




